
Carbon Nanotubes (CNTs)
In 1991 S. Iijima, while examining some sample of carbon crystals viz. fullerenes using electric arc discharge method by a transmission electron microscope, accidentally observed carbon nanotubes.
Carbon nanotubes were so less in this sample that Iijima himself stated that it was like observing ‘a needle in haystack’.
What are Carbon nanotubes?
- Carbon nanotubes are allotropes of carbon and are members of the fullerene structural family (sometimes also called buckytubes).
- These cylindrical-shaped carbon molecules have exceptional strength and stiffness.
- Nanotubes are formed by a one-atom-thick sheet of carbon called graphene, rolled to form a cylindrical hollow structure.
- In graphene, there are hexagonal rings of carbon forming a honeycomb crystal lattice in which each carbon atom is bonded to three carbon atoms with sp2 bonds.
- Diameter of nanotubes : nanometer; length : micrometer-centimeter (length-to-diameter ratio may be up to 132,000,000:1).
~Types of carbon nanotubes~
- Single-walled carbon nanotubes (SWNT): The structure of a SWNT can be conceptualized by wrapping a one-atom-thick layer of graphite called graphene into a seamless cylinder. Most single-walled nanotubes (SWNTs) have a diameter of close to 1 nanometer and can be many millions of times longer.
- Multi-walled carbon nanotubes (MWNT): Multi-walled nanotubes (MWNTs) consist of multiple rolled layers (concentric tubes) of graphene. The interlayer distance in multi-walled nanotubes is close to the distance between graphene layers in graphite, approximately 3.4 Angstrom.
There are two models that can be used to describe the structures of multi-walled nanotubes.
In the Russian Doll model, sheets of graphene are arranged in concentric cylinders.
In the Parchment model, a single sheet of graphite is rolled in around itself, resembling a scroll of parchment or a rolled newspaper.

~Structure of carbon nanotubes~
A nanotube can be formed when a graphene sheet is rolled up about the T-axis as shown in the figure below.
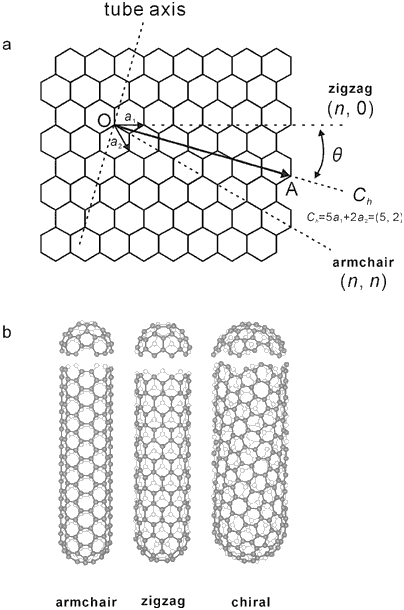
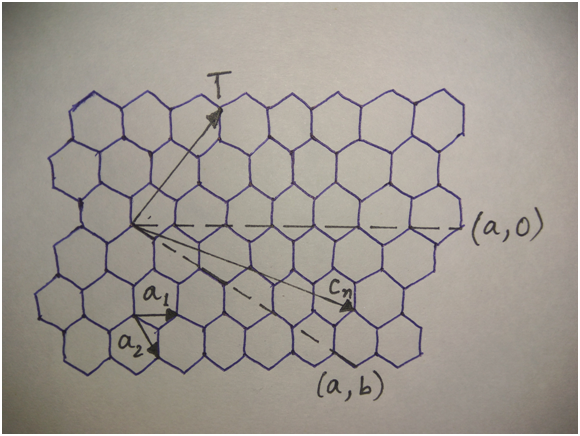
The vector Cn perpendicular to T vector is called the circumferential vector. Basically, there are three types of nanotube structures constructed by rolling the graphene sheet about T-axis having different orientation in the graphene sheet named as
- armchair structure
- zig-zag structure
- chiral structure
A vector called chiral vector describes the way the graphene sheet is wrapped. It is given by
Ch=na1 + ma2
where n and m are integers and a1, a2 denote unit vectors along two directions in the honeycomb crystal lattice of graphene.
- Armchair Structure (if m=n): When T-axis is parallel to C-C bonds of the carbon hexagons.
- Zig-Zag Structure (if n=0): When T-axis is not parallel to C-C bonds but parallel to X-axis (say). The name zig-zag has been given due to the zig-zag arrangement of carbon atoms that can be seen if the cross-section of the tube is taken. These type of tubes are ‘achiral tubes’ i.e., their mirror images are similar to the original structure.
- Helical (or Chiral) Structure (if m is not zero and n not equal to m): These are observed when T-axis makes any angle between 0 and π/6 and chirality (a,b). Such tubes are chiral and their mirror images appear to differ from their original structure.

The following table shows the comparison of three types of CNTs;
| Types of CMT | R (chirality) | Ɵ |
| Armchair (achiral) | a,a | π/6 |
| Zig-Zag (achiral) | a,0 | 0 |
| Helical (chiral) | a,b | 0 <Ɵ< π/6 |
Besides these basic types, a variety of shapes of CNTs, like ropes, strings, stripes, bamboo structure, conical shapes etc. has been observed under different experimental conditions.
~Properties of CNTs~
- Mechanical Properties: CNTs have high tensile strength and modulus of elasticity. Lighter in weight but are about 100 times stronger than steel.
- Electrical Properties: Depending on the type of CNT, these may be either metallic or semiconducting along the tubular axis. All Armchair (n=m) nanotubes are metallic while Zig-Zag and Chiral nanotubes may be metallic or semiconducting depending on the values of (n,m) and diameter.
- Thermal Properties: All nanotubes are expected to be very good thermal conductors along the tube, but good insulators lateral to the tube axis. The thermal conductivity of CNTs is very high, even higher than copper, a metal well known for its good thermal conductivity.
~Fabrication of Carbon Nanotubes~
Carbon nanotubes can be made by (i) laser evaporation, (ii) carbon arc method, (iii) chemical vapor deposition.
- Laser Evaporation: It consists of a quartz tube containing argon gas and a graphite target heated to 1200°C. The part of quartz tube outside the furnace consists of water-cooled copper collector. The graphite target contains a small amount of cobalt and nickel that act as catalytic nucleation sites for the formation of the tubes. An intense pulsed laser beam is made incident on the target, evaporating carbon from the graphite. The argon gas then sweeps the carbon atoms from the high-temperature zone to the colder copper collector on which they condense as nanotubes. Carbon nanotubes 10-20 nanometer in diameter and 100 micrometers long can be made by this technique.

- Carbon Arc Method: A potential difference of 20-25 V is applied across carbon electrodes of dimensions 5-20 micrometer diameter and separated by 1 mm at 500 torr pressure of flowing Helium. Carbon atoms are ejected from the positive electrode and form nanotube on the negative electrode. As the tubes form, the length of positive electrode decreases and a carbon deposit forms on the negative electrode. To produce single-walled nanotubes, a small amount of cobalt, nickel or iron is incorporated as a catalyst in the central region of the cathode. If no catalyst is used, the tubes are multi-walled tubes (MWCNT). The nanotubes produced by this method have the diameter in the range 0.7 nm to 2 nm and length a few micrometers. However, few millimeter long tubes have also been synthesized.

- Chemical Vapour Deposition: For large scale production of nanotubes, this method is most useful. The method consists of decomposing a hydrocarbon gas like methane (CH4) at 1100°C. As the gas decomposes, carbon atoms are produced which condense on a cooler substrate that may contain various catalysts such as iron. This method produces nanotubes with open ends which is a unique feature of this method. This procedure allows continuous fabrication and deposition of aligned nanotubes on solid substrates so that they can be used for electronic applications. Both MWCNT and SWCNT can be produced by this method. No amorphous carbon formation takes place, making high purity nanotubes. The particulars of this method are
| Gases | CH4, C6H6 |
| Pressure | >100 torr |
| Temperature | ≈1100° |
| Catalyst | Fe, Co, Ni, Pt, etc. |

~Applications~
Owing to their extraordinary thermal conductivity, mechanical and electrical properties, carbon nanotubes find applications as additives to various structural materials.
- In Composite Fibres: Because nanotubes have high tensile strength, these can be used to form composite fibers which have great strength. Such super strong fibers will have applications including body and vehicle armor, transmission line cables, woven fabrics, and textiles.
- In Structural Composites: Because nanotubes have high tensile strength and elasticity, these can be used to form structural composites.
- In Conductive Plastics: For making conductive plastics by loading plastic nanotubes.
- In Molecular Electronics (or nanoscale electronics): Their geometry and electrical conductivity make buckytubes the ideal candidates for the connections in molecular electronics. In addition, they have been demonstrated as switches themselves.
- In Thermal Conduits: Because of having high thermal conductivity, these can be used as thermal conduits.
SOURCES:
https://en.wikipedia.org/wiki/Allotropes_of_carbon
https://en.wikipedia.org/wiki/Carbon_nanotube
http://www.mdpi.com/1996-1944/3/5/3092/pdf-vor
Kailasha Foundation - Bringing Solutions To You
Follow us on Facebook, Twitter, Instagram, LinkedIn for regular updates.


Leave a reply